- Brugioni Lucio
- Brief Report and Case Report
Rapidly fatal West Nile virus meningoencephalitis in an immunocompetent patient: a case report
- 2/2019-giugno
- ISSN 2532-1285
- https://doi.org/10.23832/ITJEM.2019.022
Abstract
Background
Case Report
Conclusions
Keywords
encephalitis; meningoencephalitis; neuroinvasive disease; West Nile virus; fatal meningoencephalitis.
Introduction
Case Report
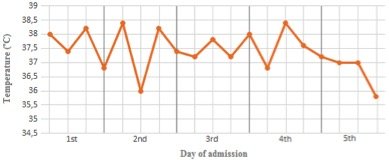
Figure 1. Patient’s temperature chart. Auricular temperature was taken four times a day (6 a.m., 12 a.m., 18 p.m., 12 p.m.)
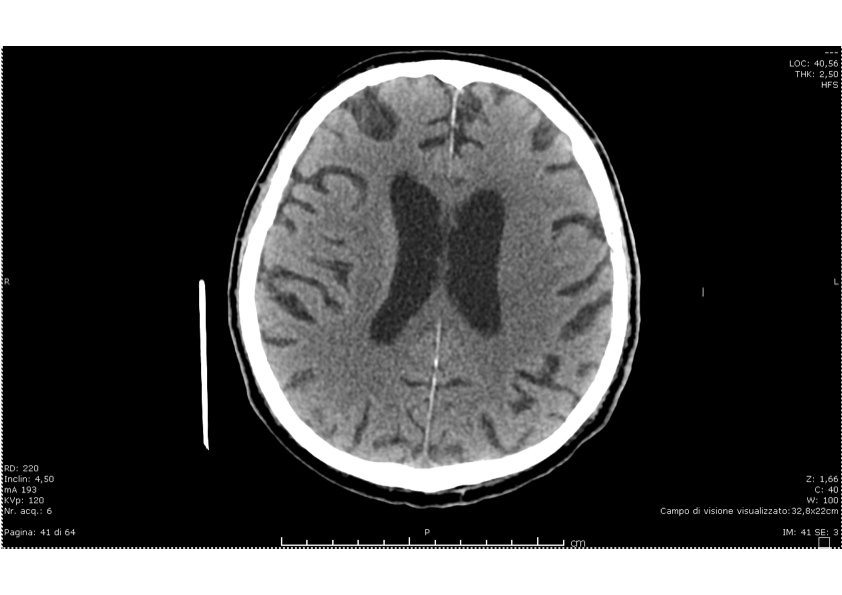
On the third day of admission the patient began lethargic with a Glasgow come score < 8.A head CT scan showed a slightly enlarged ventricular system without evidence of ischemic or hemorrhagic stroke, space-occupying lesions or asymmetry (Figure 2).
Conclusions
As underlined above, risk factors for poor prognosis are still not clearly identified to date.
A variety of factors might contribute to fatal outcome (e.g. immunosuppression states, diabetes, age) but more comprehensive epidemiologic and clinical summaries are needed. Our patient had an advanced age and suffered from diabetes mellitus and arterial hypertension. He underwent unexpected rapid clinical deterioration before any invasive treatment could be carried out. Thus, data on larger numbers of fatal WNV cases may help to promptly recognize high-risk patients who could benefit from early invasive management.
References
- Colpitts TM, Conway MJ, Montgomery RR, Fikrig E. West Nile Virus: Biology, Transmission, and Human Infection. Clin Microbiol Rev. 2012 Oct; 25(4):635–648.
- Data from World Health Organisation: http://www.euro.who.int/en/countries/italy/news/news/2018/8/west-nile-virus-infections-spike-in-southern-and-central-europe
- Groves JA, Shafi H, Nomura JH, Herron RM, Baez D, Dodd RY et al.. A probable case of West Nile virus transfusion transmission. Transfusion. 2017 Mar; 57(3pt2):850-856. doi: 10.1111/trf.14018. Epub 2017 Feb 5.
- Julander JG, Winger QA, Rickords LF, Shi PY, Tilgner M, Binduga-Gajewska I, et al. West Nile virus infection of the placenta. Virology. 2006 Mar 30;347(1):175-82. Epub 2006 Jan 9.
- Kelley RE, Berger JR, Kelley BP. West nile virus meningo-encephalitis: possible sexual transmission. J La State Med Soc. 2016 Jan-Feb; 168(1):21-2. Epub 2016 Feb 15.
- O’Leary DR, Kuhn S, Kniss KL, Hinckley AF, , Rasmussen SA, Pape WJ, et al. Birth outcomes following West Nile Virus infection of pregnant women in the United States: 2003-2004. Pediatrics. 2006 Mar; 117(3):e537-45.
- Roos KL. West Nile encephalitis and myelitis. Curr Opin Neurol 2004; 17: 343-6
- Ciota AT. West Nile virus and its vectors. Curr Opin Insect Sci. 2017 Aug; 22:28-36. doi: 10.1016/j.cois.2017.05.002. Epub 2017 May 9. Review.
- Nash D, Mostashari F, Fine A, Miller J, O’Leary D, Murray K et al. The outbreak of West Nile virus infection in the New York City area in 1999. N Engl J Med. 2001 Jun 14; 344(24):1807-14.
- Papa A. West Nile virus infections in humans—Focus on Greece. Review article. Journal of Clinical Virology, Volume 58, Issue 2, October 2013, Pages 351-353
- Rizzo C, Napoli C, Venturi G, Pupella S, Lombardini L, Calistri P, et al. West Nile virus transmission: results from the integrated surveillance system in Italy, 2008 to 2015. Euro Surveill. 2016 Sep 15;21(37).
- Bode AV, Sejvar JJ, Pape WJ, Campbell GL, Marfin AA. West Nile virus disease: a descriptive study of 228 patients hospitalized in a 4-county region of Colorado in 2003. Clin Infect Dis. 2006 May 1; 42(9):1234-40. Epub 2006 Mar 30.
- ECDC: Epidemiological update: West Nile virus transmission season in Europe, 2018 https://ecdc.europa.eu/en/news-events/epidemiological-update-west-nile-virus-transmission-season-europe-2018
- European Centre for Disease Prevention and Control (ECDC). West Nile virus infection Surveillance Atlas. Avaible from: https://ecdc.europa.eu/en/west-nile-fever/surveillance-and-disease-data/disease-data-ecdc.
- ECDC: Transmission of West Nile virus, June to December 2018 –Table of cases, 2018 transmission season. https://ecdc.europa.eu/en/publications-data/transmission-west-nile-virus-june-december-2018-table-cases-2018-transmission