- Mauro Giordano
- Original Article
Hyperchloremic metabolic acidosis as usefull tool for early suspicious of acute kidney injury in emergency department
- 2/2018-Luglio
- ISSN 2532-1285
- https://doi.org/10.23832/ITJEM.2018.020
Mauro Giordano1; Tiziana Ciarambino1; Pietro Castellino2; Lorenzo Malatino2 and Luigi Elio Adinolfi1
-
Department of Medical, Surgical, Neurological, Metabolic and Geriatrics Sciences, University of Campania “L. Vanvitelli”, Italy
-
Department of Clinical and Experimental Medicine, University of Catania, Catania, Italy
Corresponding Author’s Institution:
Mauro Giordano, M.D., Ph.D.
University of Campania “L. Vanvitelli”
Abstract
Introduction
Acute kidney injury (AKI) is a frequent and potential life-threating syndrome in emergency department (ED). We investigated on the hyperchloremic metabolic acidosis (HCMA) level in acute kidney injury (AKI) patients in ED.
Methods
In the present paper we performed a retrospective study on the HCMA level as obtained by the following arterial blood gas analysis (ABG) derived formula: ((arterial pH x arterial bicarbonate concentration) / arterial chloride concentration) and renal function evaluated by e-GFR, in both non-AKI and AKI emergency patients. We enrolled 105 patients subdivided in 2 groups. Control group (non-AKI) consisting of 55 patients with normal creatinine level and AKI group consisting of 50 patients according to KDIGO
Results
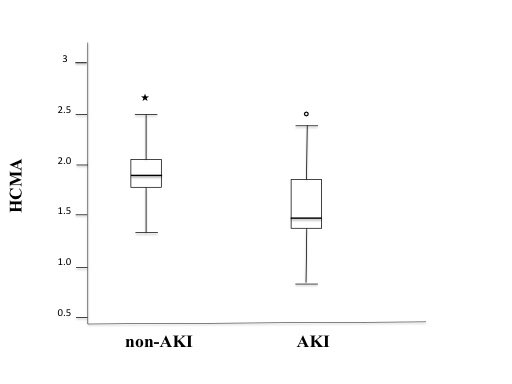
Mean creatinine level was of 0.81±0.02 in non-AKI group and it was significantly increased to 2.09±0.01 in the AKI group (p< 0.01). Mean e-GFR level was of 93.1±0.45 in non-AKI group and it was significantly decreased to 32.3±0.25 in the AKI group (p< 0.01). The mean of HCMA was of 1.88±0.09 in non-AKI group and it was significantly decreased to 1.46±0.07 in the AKI group (p< 0.001). In the AKI group we reported a significantly correlation between HCMA and e-GFR levels (r: 0.467, p < 0.01).
Conclusion
The present data suggest that HCMA level may be a useful clinical tool for the early diagnosis of acute kidney injury in emergency department.
Keywords
Acute kidney injury, hyperchloremic metabolic acidosis, emergency department.
Introduction
Previous data have reported that about 5.5% of all emergency department patients had acute kidney injury (AKI), defined as a rise in creatinine of 50% from its baseline value and/or a fall in the glomerular filtration rate (GFR) by 25%, and/or a decrease in urine output below 0.5 ml/kg/h for 6 h or more (1-3). AKI patients are associated with higher mortality. Patients with severe sepsis and septic shock exhibit a complex metabolic acidosis at intensive care unit admission, caused predominantly by hyperchloremic acidosis, which was more pronounced in non-survivors (4). Normally, the major renal defense against metabolic acidosis is the net urinary excretion of strong anions, such as chloride, and the retention of strong cations (5). This mechanism resulted impaired in AKI patients. Early diagnosis of AKI is crucial for a correct and prompt management of acute renal disfunction. Thus, the present study was performed to investigate on the usefulness of hypercloremic metabolic acidosis (HCMA) level for the early suspicion of AKI in emergency department.
Methods
Study design
We performed a retrospective study in our Emergency Department (ED). Our study was approved by the Local Ethics Committee. Inclusion criteria were: (critically hill) emergency patients with or without AKI, as defined with serum creatinine increased to ≥ 0.3 mg/dl (26.5 micrmol/L) within 48 h or 1.5-fold from the baseline, which was known or presumed to have occurred in the preceding 7 days. Exclusion criteria were: age less than 18 years, history acute gastrointestinal and post-operative patients. The Acute Physiology and Chronic Health Evaluation (APACHE II) score and Charlson Comorbidity Index (CCI) (6-7) were calculated from the patient charts. The study population included 105 patients with the following data that was obtained from each ED admission: demographic and clinical characteristics, patient diagnoses, procedures (eg, laboratory tests), treatments/interventions and patient outcomes. Patients were grouped into the non-AKI group (with normal creatinine levels and GFR > 90 ml/min/1.73 m2) and AKI group (KDIGO guidelines). In particular, AKI was diagnosed if serum creatinine increased to ≥ 0.3 mg/dl (26.5 micrmol/L) within 48 h or 1.5-fold from the baseline, which was known or presumed to have occurred in the preceding 7 days (3, 8). Data for the non-AKI group were compared with those of the AKI group. Clinical characteristics of non-AKI group and AKI group were reported in (Table 1).
Table 1
Clinical characteristics in the non-AKI and AKI groups. APACHE (Acute Physiology and Chronic Health Evaluation). The values are indicated by percentage and mean ±SE. * = p <0.01
|
non-AKI |
AKI |
|
|
Patients N. (male) |
55 (18) |
50 (21) |
|
Age (years) |
71±4 |
73±3 |
|
APACHE II score |
20±3 |
21±4 |
|
Charlson Comorbidity Index |
3.7±1.8 |
4.1±1.3 |
|
Serum Creatinine (mg/dl) |
0.81±0.02 |
2.09±0.01 * |
|
e-GFR (ml/min/1.73 m2) |
93.1±0.45 |
32.3±0.25 * |
|
Ph |
7.43±0.3 |
7.32±0.2 |
|
HCO3— |
25±8 |
21±1 |
|
Cl– |
97±1 |
108±1 |
|
pO2 |
67±3 |
61±2 |
|
pCO2 |
42±2 |
44±2 |
Measurements
Glomerular filtration rate (GFR) was evaluated by CKD-EPIdemiology Collaboration (CKD-EPI) equation (9). All acid-base variables and clinical data were collected from electronic patient charts. Serum levels of albumin, creatinine and various electrolytes, including Na, K, Ca, Mg, Cl and HPO4, were recorded for each patient. Blood pH and the partial pressures of oxygen and carbon dioxide in the blood (PaO2 and PaCO2) were measured using a RapidLab Blood Gas Analyzer (GEMr Premiertm 4000 plus). In this study we investigated on the usefulness of hyperchloremic metabolic acidosis state (HCMA), as evaluated using the following formula: ((pH x HCO3 -) / Cl -), where pH is the arterial pH, where HCO3– is the arterial bicarbonate concentration and Cl- is the arterial chloride concentration, as reported by ABG analysis, at admission to the Emergency Department.
Statistical Analysis
Data analyses were performed using SPSS, Version 19.0 (IBM Corp, Armonk, NY). Statistical significance was set at p<0.05 for all procedures. Analyses of ABG were made using a non-parametric U-test by Mann-Whitney for independent sample. Data are presented as percentage and mean ± standard error (SE).
Results
A total of 105 patients were enrolled, mean age was 72±2 years, and the clinical characteristic of patients in non-AKI and AKI groups were reported in Table 1. Mean creatinine level was of 0.81±0.02 in non-AKI group and it was significantly increased to 2.09±0.01in the AKI group (p< 0.01). Mean GFR level was 93.1±0.45 in non-AKI group and it was significantly decreased to 32.3±0.25 in the AKI group (p< 0.01) (Table 1). In particular, the mean of HCMA was of 1.88±0.09 in non-AKI group and it was significantly decreased to 1.46±0.07 in the AKI group (p< 0.01). In the figure 1 we also reported HCMA levels in AKI and non-AKI groups.
Figure 1
Discussion
In this present study using the arterial blood gas analysis (ABG) derived formula: a significant association between HCMA state and e-GFR was found in AKI patients. AKI is associated with higher mortality in emergency patients (10-11). Thus, early identification of AKI patients is crucial for a prompt management. Acute kidney injury can lead to hyperchloremic metabolic acidosis especially when the eGFR is between 30 to 59 mL/min (stage III) (12-13). To maintain normal pH, the kidneys reabsorb all the filtered HCO3– and excrete the daily H+load (14). Hyperchloremic metabolic acidosis has been reported in AKI patients (4). During the early phases of AKI, it has been reported hyperchloremic metabolic acidosis, because of impairment in renal ammonia excretion (15). In particular, in the early state of AKI there are no classic physical signs and symptoms to alert the clinician of ongoing injury. To this regard, identifying subjects with suspicion for developing AKI is crucial and the present data suggest that the ABG derived formula may have a potential as screening test to early suspect for AKI. Rapid evaluation of renal function for early management is important in AKI patients. However, at the moment, the diagnosis of AKI is usually based on changes in serum creatinine. This is a less rapid indicator of acute renal dysfunction and it may lead to a delay in the diagnosis of AKI. To this interest, we found a linear positive correlation between the level of HCMA and the impairment of renal function. This is the first report that shows this association in AKI patients. If confirmed and validated in future study, the use of ABG derived formula for HCMA may represent a useful clinical tool for the early suspicion of AKI in emergency department.
References
-
Li PK, Burdmann EA, Mehta RL. Acute kidney injury: global health alert. Kidney Int 2013; 83: 372–376.
-
Frank Xavier Scheuermeyer, Eric Grafstein, Brian Rowe, et al. The Clinical Epidemiology and 30-Day Outcomes of Emergency Department Patients With Acute Kidney Injury. Canadian Journal of Kidney Health and Disease Volume 4: 1–13 , 2017.
-
Kidney Disease: Improving Global Outcomes (KDIGO) Acute Kidney Injury Work Group. KDIGO clinical practice guideline for acute kidney injury. Kidney Int Suppl 2012; 2: 1–138.
-
Noritomi DT, Soriano FG, Kellum JA, Cappi SB, Biselli PJ, Libório AB, Park M. Metabolic acidosis in patients with severe sepsis and septic shock: a longitudinal quantitative study. Crit Care Med. 2009 Oct;37(10):2733-9.
-
Stewart P. A. “Modern quantitative acid-base chemistry,” Canadian Journal of Physiology and Pharmacology, vol. 61, no. 12, pp. 1444–1461, 1983.
-
Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 1987, 40(5):373-83.
-
Zimmerman JE, Kramer AA, McNair DS, et al. Acute Physiology and Chronic Health Evaluation (APACHE) IV: hospital mortality assessment for today’s critically ill patients. Crit Care Med 2006;34(5):1297–310.
-
Bellomo R, Ronco C, Kellum JA, et al. Acute renal failure – definition, outcome measures, animal models, fluid therapy and information technology needs: the Second International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group. Crit Care. 2004 Aug;8(4):R204-12.
-
Levey AS, Becker C, Inker LA. Glomerular filtration rate and albuminuria for detection and staging of acute and chronic kidney disease in adults: a systematic review. JAMA. 2015 Feb 24; 313(8): 837–46.
-
Hoste EA, Bagshaw SM, Bellomo R et al. Epidemiology of acute kidney injury in critically ill patients: the multinational AKI-EPI study. Intensive Care Med 2015; 41: 1411–1423.
-
Bouchard J, Acharya A, Cerda J et al. A prospective international multicenter study of AKI in the intensive care unit. Clin J Am Soc Nephrol 2015; 10: 1324–1331.
-
Bockenkamp B and Vyas H. Understanding and managing acute fluid and electrolyte disturbances. Current Paediatrics 13: 520-528, 2003.
-
Relman AS, Lennon EJ, Leman J. Endogenous production of fixed acid and the measurement of the net balance of acid in normal subjects. J Clin Invest 40; 1621, 1961.
-
Widmer B, Serum electrolyte and acid base composition. The influence of graded degrees of chronic renal failure.Gerhardt RE, Harrington JT, Cohen JJ. Arch Int Med 139:1099,1979.
-
Wright G, Noiret L, Olde Damink SWM, Jalan R. Interorgan ammonia metabolism in liver failure: the basis of current and future therapies. Liver Int, 2011; 31: 163–75. 147.