- Maria Barbara Schito
- Brief Report and Case Report
Multidisciplinary approach to intravascular leiomyomatosis with cardiac interesting (intracardiac extension)
- 1/2018-Febbraio
- ISSN 2532-1285
- https://doi.org/10.23832/ITJEM.2018.010
M. B. Schito1, G.M. Colombo1, G. Aguglia1, G. Carlotti1, L.R. Del Vecchio1, F. Pace1, A. Tavanti1, M. Vitto1, F. Ranocchi2, P. Le Piane3, D. Cartoni4, E. Guglielmelli1.
Abstract
Case Presentation
Conclusions
Introduction
Leiomyoma is a histologically benign tumor of the smooth muscle cells. The intravenous leiomyomatosis is an unusual clinical condition, characterized by intravascular proliferation through the lumen of the iliac veins to the inferior vena cava (VCI) with extension to the right hearth. This clinical condition is called IVL (1,2,3). In 1959, for the first time, IVL was described in English literature from Marshall e Morris and, since than until 2012, they have been reported 194 cases (4). In 1984, the first surgery has been described by Darks (5,6,7,8,9). The higher incidence is in women between 40 and 50 years, in pre-menopausal age; most patients (approximately 83,5%) had undergone a previous hysterectomy/myomectomy (3). Only a few patients have normal uterus and this supports possible origin of the smooth muscle cells of the tunica media of the vessel wall.
The non-specific symptomatology is correlated with the severity of cardiac involvement. The IVL can be related to the obstructive effect on the tricuspid orifice and disturbance of venous return into the right atrium, which could include pulmonary dyspnea, syncopal episodes, pulmonary embolism and sudden death. Differential diagnosis include atrial mixoma, inferior vena cava thrombosis, abdominal tumors such as hepatocellular carcinoma, renal cell carcinoma, Wilms tumor (typical of children), adrenal tumor, leiomyosarcoma of the uterus and metastatic tumors (10,11).
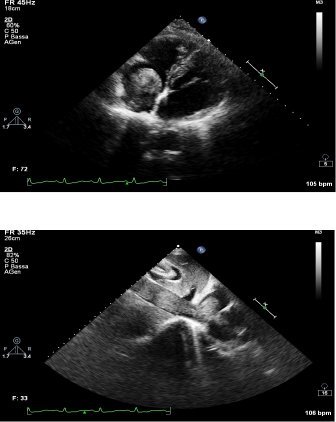
Atrial mixoma originates from the interatrial septum and extended to the left ventricular cavities; in preoperative step, kidney cancer can distinguished with imaging that can highlights kidney lesion, but in postoperative step, kidney cancer can diagnosed with histological examination. “Imaging” has allowed a correct preoperative diagnosis. Trans-thoracic echocardiography (TTE) can give a “real-time” information on the intra-cardiac lesion, its size and mobility. Trans- esophageal echocardiography (TEE) revealed in a more detailed way a hyperechoic and mobile mass that filled until 2/3 of the right atrium and protruded across the tricuspid valve (12,13). The contrast- enhanced CT and MRI are used to define sites, localization, origin and extent and to give accurate anatomical information (14,15).
Surgery can be performed in one or two stages: resection of intrathoracic and abdominal/pelvic tumor in two separate operations or involving total resection of the tumor (16,17,18,19). It is decided on the basis of the clinical condition of the patient and of the surgical agreements that may change during surgery for the appearance of complications. Surgery with complete excision of the tumor is strongly recommended for a favorable outcome; no recurrent disease has been reported in 15 years follow-up.
Histologic evaluation of IVL is characterized by uniformity with fasciculations or plexiform appearance or occasionally storiform without atypia or increased mitotic index (5). Immunohistochemical analysis using monoclonal antibodies revealed positivity for smooth muscle markers desmin and smooth muscle actin (SMA) and optionally receptors for estrogen and/or progesterone. The proliferative index Ki-67 is minimal (<1%). Treatment with antiestrogens or aromatase inhibitors is very controversial, and it was considered only for patients with partial resection of disease.
Case Presentation
In september 2014, a 47 years-old woman, nulliparous, in premenopausal, was submitted to myomectomy. In april 2016, for local recurrence, in the Department of Gynaecology, she has been subjected to subtotal hysterectomy with preservation of annexes. Histological examination confirmed leiomyomatosis with endovascular thrombi and ischemic necrosis. In December 2016, the patient was hospitalized for shortness of breath, bilateral legs edema and simultaneously increase volume of abdomen.
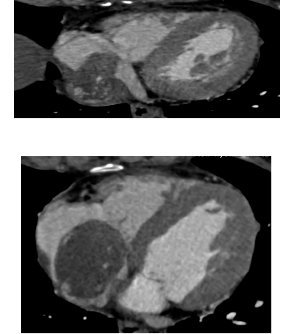
Abdominal CT documented “polilobata”, inhomogeneous and vascularized pelvic tumor mass, that compressed the surrounding structures, like the bladder, without any cleavage plane, and that determined bilateral hydroureteronephrosis, predominantly to the left; furthermore, CT showed a long, serpentine and elongated mass extending from internal left iliac vein continuing to the inferior vena cava and extending into the right atrium (Fig. 1). This long mass obliterate, almost completely, the lumen of the intrahepatic cava and right atrium and partially the lumen of left renal vein and right iliac veins.
Initially, origin of this mass was unknown; likely referring to thrombus extending from iliac veins to right atrium. The surgical remove of the pelvic mass was subordinated to the resolution of the “thrombotic” issue. Meantime, in the light of bilateral “idroureteronefrosi”, a double bilateral JJ urethral stent was placed; at the same time, sodium heparin therapy was started and changed to low molecular weight heparin for approximately 45 days, without any clinical benefit.
The integration of other diagnostic tests allowed a correct diagnosis. TTE (Fig. 2) showed voluminous oval mass approximately of 28×42 mm, that filled the right atrium and that was a part of a elongated, hypoecogenic and inhomogeneous mass (20-25 mm in diameter). This elongated mass filled the lumen of the VCI, and its thrombotic nature are not likely, in the light of the poor response to anticoagulant therapy, but was compatible with intravascular leiomyomatosis. Chest CT confirmed the presence of solid tissue, with the same densitometric characteristics of the pelvic mass, that filled and dilated completely the left internal iliac vein and the VCI and extended to the right atrium (diameter of 5×3,5 cm).
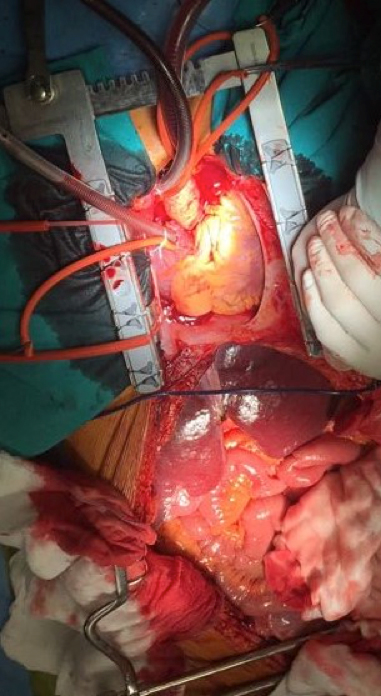
First of all, the patient was undergo embolization of uterine arteries. At a later time surgery has been planned in two steps. In the first part (Fig. 3), after sternotomy in extracorporal circulation and hypothermia (26°C), the cardiac surgeon has removed, opening the right atrium, the coarse neoformation with smooth marigins that projected into the right atrium. It was necessary a longitudinal venotomy of the inferior vena cava in its thoracic part (Fig. 4). During the same operation, through midline laparotomy, the general surgeon performed an initial cavotomy to extract the segment of thoraco-abdominal neoformation and, after the clamping of the left common iliac vein, he removed the infrarenal segment of the neoformation strongly adherent to the IVC wall. It was possible to visualize the pelvic mass that attracted the bladder and the sigmoid, twisted on itself, and to observe new local venous hypertension vessels.
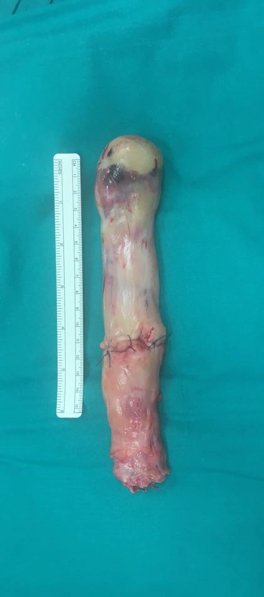
Following the prolonged hypothermia, time for extracorporeal circulation, and considering the numerous pelvic collateral circles, it has been decided to postpone the second part of the surgery by removing the pelvic mass and the IVL in the left internal iliac vein. After seven days, the patient has been subjected to laparotomy to remove “en Bloc” the tumor and both ovaries and appendages. The uterus neck, that appeared dislocated to right and the residual IVL in the left internal iliac vein has been removed.
This neoformation incorporated the intrapelvic tract of the left ureter; this tract has been removed and the upstream and downstream moncons was sutured, leaving the “double JJ” urethral stents previously positioned. In this way, strongly related to the bladder, the pelvic mass has been completely removed.
The hypogastric artery and the vein have been linked and sectioned to perform a complete and radical removal of the IVL. The extemporaneous histologic examination showed bundles of isolated spindle cells and necrosis areas. The final hystologic examination is compatible with leiomyomatosis intravascular that originate from uterus and confirmed fusiform cells with elongated nuclei to smooth cells origin. On the immunohistochemical study, the tumor cells were positive for actin, desmin, CD34, with mild nuclear atypia and absence of mitosis; there was no positivity with the antibodies for S-100 protein, CD 117 and CD10. This panel showed many vessels, some of this containing thrombi, and associated with ischemic necrosis; the proliferative index (percentage of positive tumor cells to Ki-67) was 7%.
Figure 1. Thoracic CT scan. It should be noted the characteristic fusiform appearance of the tumor that grows in the vena cava until complete protrusion into the right atrium.
Conclusions
The IVL is a rarely encountered smooth muscle cell tumor that originated from the uterus and grows within venous channels but does not invade the tissues. Occasionally, the tumor occurs in other intravascular locations, distant from the uterus. Intracardiac leiomyomatosis is insidious; the patient can develop heart failure, pulmonary embolism and sudden death. The diagnostic tests, trans-thoracic and/or trans-esophageal echocardiography, computerized tomography and/or MRI allows an accurate diagnosis on origin, on intravascular extension and localization.
The gold standard treatment is a surgical complete removal, with hysterectomy and resection of all extra-uterine masses, mandatory for a favorable outcome and to avoid recurrence. Surgery can be performed in one or two stages, with resection of the intrathoracic tumor–intravascular and intacardiac tumor in hypothermia, cardiac arrest and extracorporeal circulation, to reduce the risk of death from obstacle to cardiac inflow, from obstruction of the tricuspid valve or from pulmonary embolism- and of the abdominal/pelvic tumor in two separate operations. Hence, surgery is really curative.
The tumor recurrences are uncommon and occur particularly when complete removal has not been possible for several reasons. Bilateral oophorectomy and removal uterus broad ligament (possible additional site of leiomyomatosis) are also considered essential, because this tumor is estrogen-dependent. The affinity for the intravascular growth could be associated with the histological positivity for estrogen and progesterone receptors. In cases of unresectable residual tumor, anti-estrogens therapy, such as tamoxifen, has been considered but efficacy is still controversial.
Long-term prognosis and survival of IVL is usually very good if the disease can be completely removed. Recurrence are unusual but can be found up to 15 years after surgery. Long-term follow-up is recommended, including thorax and abdomen CT scan and serial echocardiography.
References
- Ryan J.Carr, M.D, Pei Hui, M.D., Ph.D., and Natalia Buza, M.D. Intravenous Leuiomyomatosis Revisited: an Experience of 14 cases at a single medical center. International Journal of Gynecological Pathology 34:169-176, Lippincott Williams & Wilkins, Baltimore 2015 International Society of Gynecological Pathologists
- Clement PB. Intravenous leiomyomatosis of the uterus. Pathol Annu 1988; 23 (pt 2): 153-83
- Cornelis F., Belleannee G., Lederlin M. Cardiac extension of an intravascular leiomyomatosis 43 years after hysterectomy. Thorac Cardiovasc Surg 2012; 144: e3-5.
- Bin Li, Xin Chen, Ya-Dong Chu, Ren-Yuan Li, Wei-Dong Li and Yi-Ming Ni. Intracardiac leiomyiomatosis: a comprehensive analysis of 194 cases. Interactive Cardiovascular and Thoracic Surgery 17 (2013) 132-139
- Cigdem Vural, Ozlem Ozen, Beyhan Demirhan. Intravenous lipoleiomyomatosis of uterus with cardiac extension: a case report. Pathology-Research and practice. Department of Pathology, Baskent University Faculty of Medicine, Ankara, Turkey, 2010
- K. Wu, Intravenous leiomyomatosis with intracardiac extension, Internal Medicine 48 (2009) 997-1001
- A. Andrade, Intravenous leiomyomatosis of the uterus: a report of three cases, Pathology Oncology Research 4 (1998) 44-47
- Lou at al. Intravenous leiomyomatosis of the uterus with extension to the right heart. Cardiovascular Ultrasound 2011; 9:25
- Durck H. Ueber ien Kontinvierlich durch die entere Holhlvene in des Herz vor Wachsendy: fibromyon des uterus. Munchen Med We-hnschr 1907; 54:1154
- Ines Cruz, Isabel Joao, Bruno Stuart, Mario Iala, Luisa Bento, Carlos Cotrim, Angelo Nobre, Helder Pereira. Intravenous leiomyomatosis: a rare cause of intracardiac mass. Rev Port cardiol 2014; 33(11): 735 e 1-735.e.5
- Leitman M., Kuperstein R, Medalion B, et al. A highly unusual right atrial mass presented in two women. Eur J Echocardiogr. 2008; 9: 8333-834
- Rongjuan Li, Yanguang Shen, Yan Sun, Chuanchen Zhang, Ya Yang, Jiao Yang, Ruijuan Su, Bo Jiang. Intravenous Leiomyomatosis with Intracardiac extension: Echocardiographic Study and Literature Review Surgical Technique
- Yu-Feng Lou, Xin-Ping Shi, Ze-Zhou Song. Intravenous leiomyomatosis of the uterus with extension to the right heart. Cardiovascular Ultrasound 2011, 9; 25
- Hanjiang Zeng, Zhongzi Xu et al. Intravenous leiomyomatosis with intracardiac extension depicted on computed tomography and magnetic resonance imaging scans: a report of two cases and a review of the literature. Oncology letters 11 (2016): 4255-4263
- Zhi-Feng Xu, Fang Yong, Ying-Yu Chen, Ai-Zhen Pan. Uterine intravenous leiomyomatosis with cardiac extension: Imaging characteristic and literature review. World J of Clin Oncol 2013 10; 4(1) 25-28
- Lei Yu et al. Intravenous Leiomyomatosis with intracardiac extension: a report of two cases. J Card Surg 2011; 26:37-68
- Natalie C. Moniage, Leslie M. Randele. Uterine leiomyomatosi with intracaval and intracardiac extension. Gynecology Oncology Report 2 (2012); 130-132
- Xihui Li, Feng Xiao, Yinmo Yang, Yindong He and Siyu Zhang. One-stage complete resection of giant intracardiac leiomyomatosis with moderate hypothermia extracorporeal circulation and beating heart technique with 36 months follow-up-a case report. Journal of Cardiothoracic Surgery (2016) 11; 64 10
- Marcia Maria Morales, Alexandre Anacleto, Joao Carlos Leal, Sergio Carvalho, Jeronimo Del’Arco. Intravascular leiomyoma with heart extension. Case report. Clinics 2012; 67 (1):83-87