- Antonio Polenta
- Brief Report and Case Report
Acquired hemophilia A: a case report
- 3/2017-Ottobre
- ISSN 2532-1285
- https://doi.org/10.23832/ITJEM.2017.021

Abstract
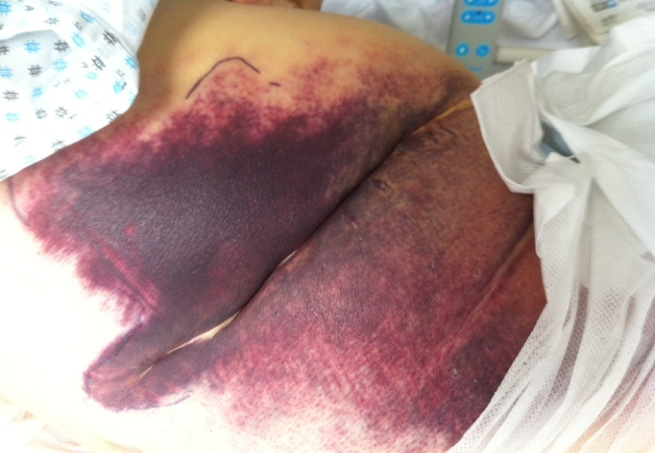
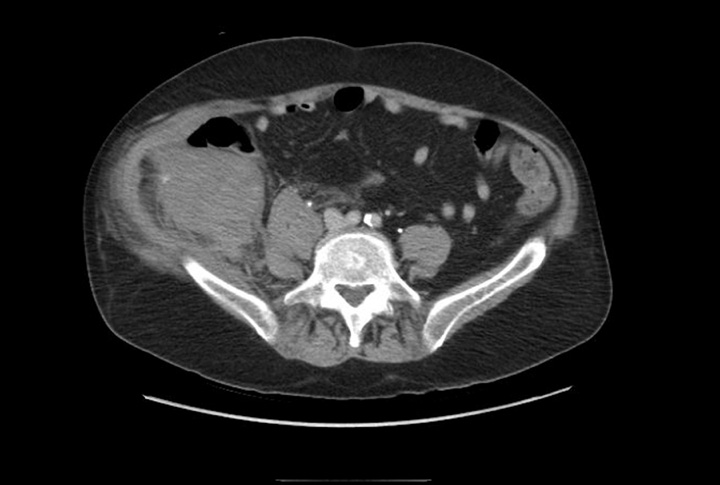
Case Report
| DAY |
aPTT (sec)
|
| 1 | 56 |
| 2 | 53 |
| 3 | 56 |
| 4 | 49 |
| 5 | 54 |
| DAY | aPTT (sec) |
| 6 | 48 |
| 7 | 50 |
| 8 | 40 |
| 9 | 44 |
| 10 | 51 |
| 11 | 48 |
| 12 | 48 |
In conclusion, why did he suddenly developed bleeding?
- the use of rFVIIa or activated prothrombin complex concentrate (aPCC) are recommended for the treatment of severe bleeding in patients with AHA.
- rFVIIa dosage: bolus injection of 90 mcg/kg every 2-3 h until hemostasis is achieved.
- aPCCs dosage: bolus injection of 50-100 IU/kg every 8-12 h (daily maximum dose of 200 IU/kg/day).
The optimal therapeutic strategy for AHA inhibitor eradication has not yet been defined, but for the last two decades, immunosuppressive regimens applied most successfully to suppress autoantibody production have included corticosteroid therapy alone or in combination with cyclophosphamide. Recently, rituximab has emerged as a promising new agent for the eradication of inhibitors in patients with acquired hemophilia. Initial treatment should therefore usually consist of corticosteroids, either alone or in combination with cyclophosphamide, especially if the patient has already been treated with corticosteroids for other medical conditions. If no response is observed after 4-6 weeks, treatment with rituximab alone or in combination with corticosteroids is an alternative. It recommended that all AHA patients be treated initially with corticosteroids alone or in combination with cyclophosphamide to eradicate autoantibodies. Suggested steroid therapy for inhibitor eradication is 1 mg/kg/day PO for 4-6 weeks, either alone, or in combination with cyclophosphamide at a dose of 1.5-2 mg/kg/day for up to six weeks. In essence the goal is to control bleeding during the time that it takes for the immunosuppressive therapy to reduce production of anti FVIII auto antibodies. If this is not done prognosis will be poor. The bleeding captures our attention but it is the immunosuppression that will save the patient. The time needed to attain this is measured more often in weeks rather than days. The use of aFVII is based on its use in hemophiliacs that have developed allo antibodies to FVIII. The rational is that a huge amount of aFVII will simply by-pass the deficient FVIII. The problem with aFVII is the doses recommended are in fact huge and difficult to procure in the amounts needed. It may be necessary to use other more easily available products after the hyper acute phase is over.
References
- International recommendations on the diagnosis and treatment of patients with acquired hemophilia A. Angela Huth-Kühne, Francesco Baudo,Peter Collins,Jørgen Ingerslev, Craig M. Kessler, Hervé Lévesque,Maria Eva Mingot Castellano, Midori Shima, and Jean St-Louis.
- Grunewald M, Beneke H, Guthner C, Germowitz A, Brommer A, Griesshammer M. Acquired haemophilia: experiences with a standardized approach. Haemophilia 2001;7:164-9.
- Ma AD, Carrizosa D. Acquired factor VIII inhibitors: pathophysiology and treatment. Hematology Am Soc Hematol Educ Program 2006:432-7.