- Stella Ingrassia
- Brief Report and Case Report
Early detection of systemic embolism: usefulness of bedside echocardiography in the emergency department
- 2/2017-Giugno
- ISSN 2532-1285
- https://doi.org/10.23832/ITJEM.2017.005
S. Ingrassia1, O. Cutuli2, L. Di Odoardo3, A. Valbusa4
- UOC Pronto Soccorso e Medicina d’Urgenza, IRCCS Fondazione Ca’ Granda Ospedale Maggiore Policlinico, Milano
- UOC Pronto Soccorso e Medicina d’Urgenza, IRCCS AOU San Martino-IST, Genova
- UOC Malattie Cardiovascolari Fondazione IRCCS Ca’ Granda Ospedale Maggiore Policlinico, Dipartimento di Scienze Cliniche e di Comunità, Università degli Studi di Milano
- UOC Cardiologia, IRCCS AOU San Martino-IST, Genova
Abstract
Renal artery obstruction is a complication of systemic thromboembolism that, if early diagnosed, can be treated with endovascular options. We report a case of renal artery embolism in a patient with mitral valve stenosis and atrial flutter. We would like to emphasize the importance of oral anticoagulation in patients with valvular atrial fibrillation and atrial flutter and the significance of bedside transthoracic echocardiography to detect possible causes of systemic embolism. Although transthoracic echocardiography sensitivity in the detection of systemic thromboembolism sources is lower than that of transoesophageal echocardiography ,bedside echocardiography is a useful, fast point of care tool which should be applied to all ED patients who present with an incresed risk of thromboembolic events.
***
A 69 years old woman referred to Emergency Department (ED) complaining of right flank and abdominal pain, dizziness and vomiting. She reported history of mitral valve commissurotomy due to rheumatic mitral valve stenosis about 30 years ago. Since then no more cardiologic follow-up was performed.
She was treated with aspirin, PPI and painkillers for osteoarthritis.
At admission her vital signs were normal. Clinical examination revealed abdominal tenderness in epigastrium and right upper quadrant. Giordano sign was positive on the right side. Laboratory results showed leukocytosis and inflammation (CRP: 160 mg/dl, normal values <5 mg/dl). ECG showed atypical atrial flutter with 4:1 conduction and normal heart rate (75 bpm). Althoug her CHADSVASC score was:2 and despite a low risk of bleeding (HAS-BLED:1) she was not on anticoagulation therapy.
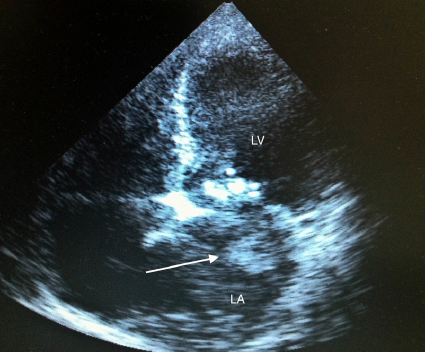
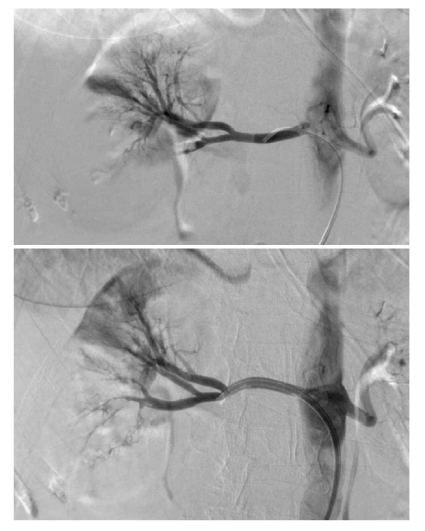
Bedside echocardiography was performed and showed severe mitral stenosis with left atrium enlargement and evidence of a hyperechogenic mobile mass , (Figure.1). A TT echocardiography executed 4 days before, did not describe such findings . A cardioembolic event due to left atrial thrombosis was then suspected. Abdominal CT was performed and diagnosis of right kidney infarction due to embolic occlusion of distal right renal artery was done. The patient was immediately transferred to Interventional Radiology Unit, renal arteriography and thromboaspiration were performed, with complete recovery of kidney perfusion.(Fig.2). The patients was first transferred to Emergency Medicine Subintensive Unit where anticoagulation with UFH was started and a 36 hours monitoring was done. The patient was then transferred to the Intensive Cardiac Care Unit (ICCU)
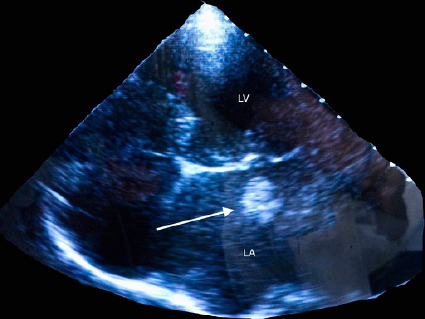
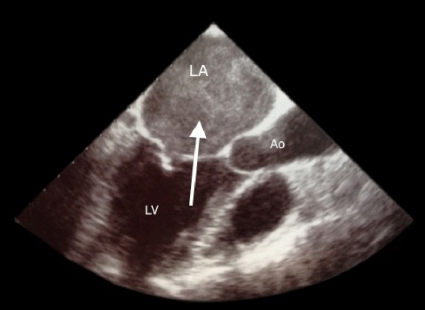
Follow-up transthoracic echocardiography confirmed the previous findings of mitral valve stenosis with pulmonary hypertension (PAPS: 50 mmHg), severe tricuspidal regurgitation, and left atrial thrombosis (Fig.3). Transesophageal Echocardiography performed after 60 hours of effective anticoagulation revealed complete resolution of left atrial thrombosis (Fig.4).
Six days after ED access, mitral valve replacement with bio-prosthesis and tricuspid valvuloplasty were performed. After 19 days from ED access the woman was discharged in good clinical conditions and warfarin was carried on before discharge, vitamin K antagonists were judged the best choice since valvular atrial fibrillation was present . [1]
DISCUSSION
Renal artery embolism (RAE) is a rare condition, its reported post-mortem incidence is around 1.4%, but its clinical significant incidence is about 0.007% of hospitalized patients. [2]
Most common risk factors for RAE include atrial fibrillation, ischemic heart disease, mitral valve disease, previous embolism and aortic valve disease [3-4-5-6], though other aetiological factors include trauma, polycythemia vera, fibromuscolar dysplasia, dissection of aorta or renal artery and extraadrenal pheocromocytoma, [7]. The most common symptom is abdominal, flank or back pain; vomiting and fever are also frequent, as in our case. Leucocytosis is often present, serum creatinine is usually elevated, also due to frequent exposure to contrast during diagnostic workup. Diagnosis is usually made by Computed Tomography, which has a much higher sensitivity than ultrasound, though the gold standard diagnostic test is renal angiogram [2-8].
Our case reminds the importance of anticoagulation in patients with paroxysmal or permanent atrial fibrillation regardless of the maintenance of sinus rhytm and in particular in case of mitral stenosis (MS) [9]. Althoug our patient presented a high risk for cardioembolic stroke (CHA2DS2VASC score of 2 in presence of mitral stenosis) and despite a low risk of bleeding (HAS-BLED:1) she was not on anticoagulation therapy. We remind that in patients with mitral valve stenosis anticoagulation is indicated even if sinus rhythm is present where there has been prior embolism, or a thrombus is present in the left atrium (reccomendation Class I, level C) [9].
Our patient, treated with aspirin, developed several years after commisurotomy, a relapse of severe mitral stenosis.This situation, probably determining left atrial enlargment and pulmonary hypertension, favoured atrial flutter and consequent left atrial thrombosis. After prompt diagnosis and a 60 hours effective anticoagulant treatment left atrial thrombosis disappeared.
Today bedside ultrasonography plays an important role especially in the emergency setting.
During emergency evaIuation, as in our case, it is crucial to obtain an actual echographic image using bedside ED ultrasonography, even if the patient has performed normal very recent exam.The utility of transthoracic echocardiography to identify or exclude left atrial or atrial appendage thrombi generally is limited, with a reported sensitivity of 40 to 60 percent, due to poor visualization of the left atrial appendage, where most atrial thrombi reside. It is known that TEE was 93 percent sensitive and 100 percent specific for thrombus, whereas the sensitivity of transthoracic echocardiography was only 53 percent [10]. In one series of patients with rheumatic mitral stenosis left atrial thrombi detected with TEE were approximately 33 percent [10]. In our case we were able to detect with a bedside TT echocardiography executed in the emergency room, a giant left atrial thrombus even if a recent echocardiogram previous excluded intracavitary thrombus or spontaneous echo contrast.With a prompt diagnosis , we were able to properly address the patient to the ICCU where she could complete her diagnostic and therapeutical process. Bedside echocardiography is a useful, fast point of care tool which should be applied to all ED patients who present with an incresed risk of thromboembolic events and its utility as a routinary point of care exam in the Emergency Department has already been investigated [11].
Figure 1: Bedside echo modified four-five chambers view, note giant left atrial mass(arrow), left atrial enlargment and mitral calcifications.
LA:left atrium LV: left ventricle
Figure 2: renal artery CT-angiography showing complete recovery of kidney perfusion after thromboaspiration
Figure 3: standard Transthoracic echocardiography four chambers view: giant floating left atrial thrombus (arrow), left atrial enlargment and mitral calcifications
LA:left atrium LV: left ventricle
Figure 4: Transesophageal Echocardiography performed after 60 hours of effective anticoagulation note complete resolution of left atrial thrombosis with persistent smoke and spontaneous echo contrast (arrow).
LA:left atrium LV: left ventricle Ao:aorta
None of the authors have conflict of interests about this case record.
References
- “2012 focused update of the ESC Guidelines for the management of atrial fibrillation”, European Society of Cardiology, 2012
- Babatunde Komolafe et al, “Successful aspiration and Rheolytic thrombectomy of a renal artery inferct and review of current literature”, canadian journal cardiology, 2012
- Kansal et al. “Renal Artery Embolism”, J Gen Intern Med 2007
- Lessman RK et. al, “Renal artery embolism: clinical features and long term follow up of 17 cases”, Ann Intern Med 1978
- Domanovitis H et al., “Acute renal infarction. Clinical characteristics of 17 patients”, Medicine 1999
- Korzets Z et al., “The clinical spectrum of acute renal infarction”, Isr Med Assoc J, 2002
- Yavuzgil et al, “Bilateral Renal Arterial Embolisation in a Patient with Mitral Stenosis and atrial fibrillation: an Uncommon Reason of Flank Pain”, Anadolu Kardiyol Derg, 2003
- Kansal S., Renal artery embolism. A case report and review. J Gen Intern Med 2008
- Guidelines on the management of valvular heart disease (version 2012), European Society of Cardiology, 2012
- Scott D. Salomon, “Essential Echocardiography – A Pratical Handbook With DVD”, 2007, Totowa, New Jersey
- Riley D, “Emergency Department diagnosis of mitral stenosis and left atrial thrombus using bedside ultrasonography”, Acad Emerg Med, 2010
Acknowledgement
None